Spike protein trimer, subdomains and variants
Coronaviruses use the S or Spike proteins, which form the characteristic spicules on their exterior, to bind to their target in our body, the cellular protein called angiotensin II converting enzyme or ACE2. For this reason, Spike is considered the key to the virus's access to the cell and constitutes the most important tool for studying and attacking the virus as it is considered the immunodominant antigen.
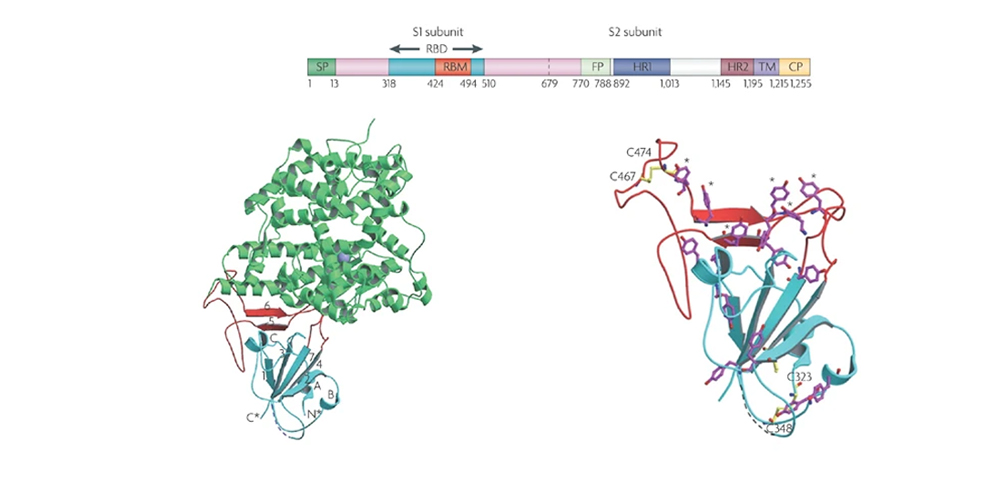
The Spike (S) glycoprotein or spicule is a viral fusion protein found in the outer envelope of the virion and plays a critical role in viral infection. It recognizes the receptors of the human cell and mediates the fusion of the viral and cellular membranes, which allows the virus to enter the cell. In structural terms, the spike (S) transmembrane glycoprotein forms homotrimers protruding from the viral surface, it contains an S1 subdomain, responsible for binding to the host cell receptor, and an S2 subdomain, which facilitates the fusion of the viral and cellular membranes. The S1 subdomain includes the receptor-binding domain (RBD) which is today the element most studied as a vaccine, therapeutic target and diagnostic tool, since it contributes to the stabilization of the membrane-anchored S2 subdomain, which contains the apparatus of fusion. S is then cleaved by host proteases at the S2 ′ site located immediately adjacent in sequence to the fusion peptide which is believed to activate the protein for membrane fusion through irreversible structural changes.Protein S has become an important target for vaccines, therapeutic antibodies, and diagnostic tests, and is required for clinical, biological, biochemical, epidemiological, virological, genetic, and structural investigations.
Our protein has been tested as an antigen in quantitative diagnostic assays and as an immunogen for the generation of therapeutic sera, with excellent results. In response to the new challenges posed by the pandemic, our team has developed a wide range of products that include the S1 and RBD subdomains, the full length Spike proteins of the main variants of epidemiological interest and the ACE2 receptor.